Clinical Research
Since its establishment in 2002, MUWRP has positioned itself as a leader in biomedical research in Uganda, pioneering efforts to combat some of the world's most deadly diseases.
Our Studies
MUWRP has successfully conducted a diverse array of groundbreaking studies, including
1. Clinical Trials
Since its establishment in 2002, MUWRP has positioned itself as a leader in biomedical research in Uganda, pioneering efforts to combat some of the world's most deadly diseases.
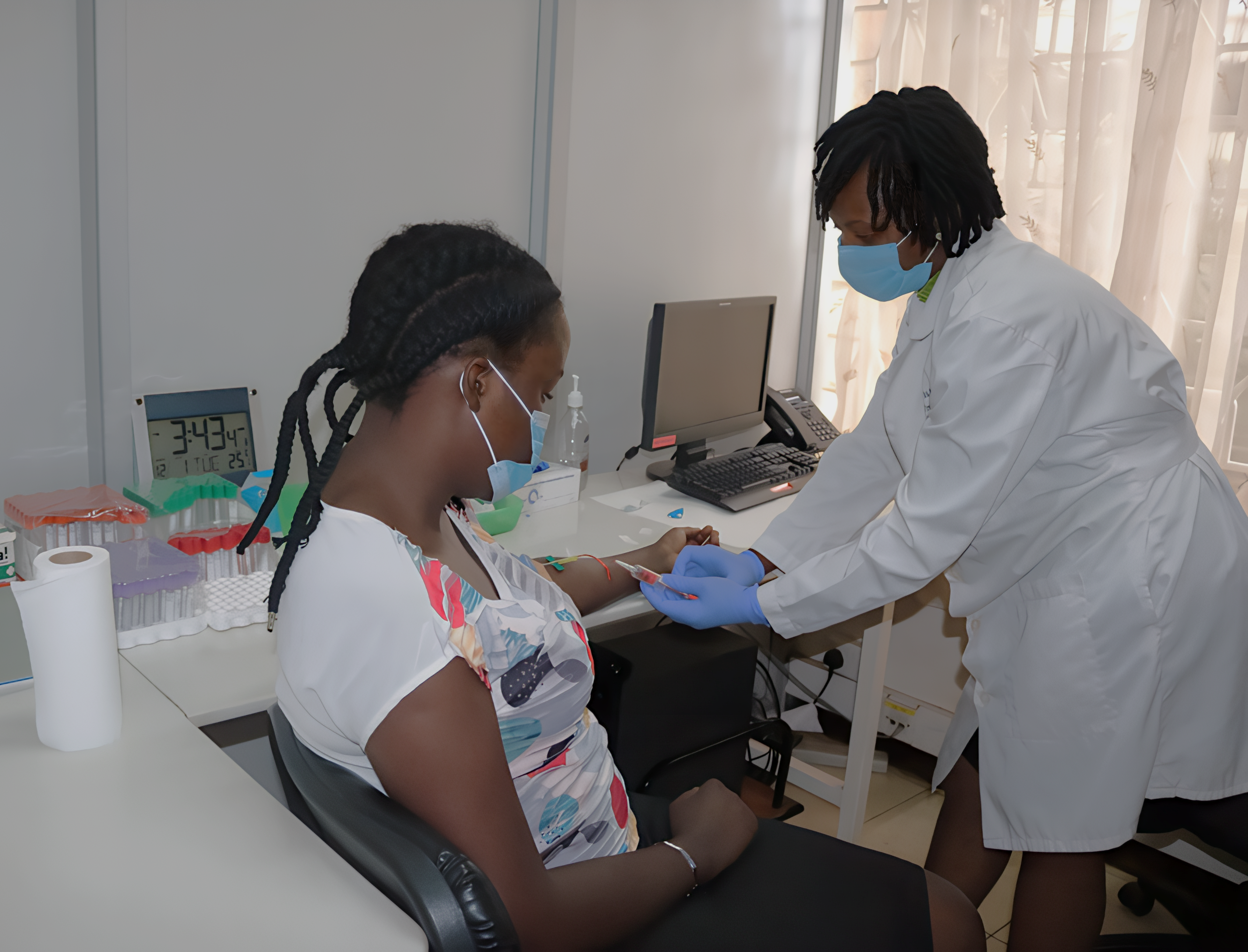
✔️ HIV Vaccine trials:Since 2024, we have conducted five Phase I/II HIV vaccine trials, testing various vaccine products and platforms such as DNA, protein subunits, and viral vectors (including Ad5, Ad26, Chimpanzee Adenovirus, and MVA vectors).
✔️ Ebola Vaccine trials: We have conducted five Phase I/II Ebola vaccine trials, including Africa’s first Ebola vaccine trial in 2007. Two trials administered candidate vaccines to vulnerable populations including HIV infected participants and children and adolescents.
✔️ Schistosomiasis Vaccine development: In 2019, we initiated Africa’s first Phase I trial to identify the dose and assess safety and efficacy of a candidate vaccine containing recombinant Sm-TSP-2.
✔️ Cohort Studies: – including high risk individuals followed twice weekly to detect acute HIV infections, in early stages Fiebig 1 and 2 and providing critical insights for cure research. The African Cohort Study (AFRICOS) is a multisite and multinational observation study following up HIV positive individuals on ART and providing data to evaluate the treatment programs and inform management policies.
✔️ Acute HIV treatment trials: Our team has conducted trials administering monoclonal antibodies with immediate or delayed ART in participants with acute HIV infection, supported by NIH DAIDS.
✔️ HIV Cure Research: We have conducted studies characterizing HIV immunity and reservoirs as part of efforts to develop a cure, also funded by NIH DAIDS.
✔️ Marburg Vaccine trials: our team has conducted early phase vaccine trials assessing safety and immunogenicity of Marburg vaccines. Phase Ib assesses the DNA Plasmid Vaccine in combination with an Ebola DNA Plasmid Vaccine as while a Phase II assessed the Monovalent Chimpanzee Adenoviral-Vectored Marburg Virus Vaccine.
2. Research Infrastructure
Our research activities are supported by a College of American Pathologists (CAP)–accredited laboratory, which operates in full compliance with internationally recognized standards. This accreditation ensures high levels of quality, accuracy, and reliability in laboratory testing, providing a strong foundation for credible research, clinical studies, and data generation across MUWRP programs
3. Continuous Quality Improvement
We implement rigorous continuous quality improvement processes guided by a comprehensive Quality Management Plan. This framework ensures that all activities adhere to the highest standards and guarantees the collection of high quality, ethical, and credible data. Through regular monitoring, evaluations, and corrective actions, we promote consistency, accountability, and continuous learning across programs to support reliable research.
Our Capabilities and Impact
✍🏽 Strong Collaborations and Trust:
Our commitment to high-quality trial execution has fostered long-term collaborations and earned trust within the research community, attracting new partnerships and funding opportunities.
✍🏽 Research Infrastructure – We have multiple research sites strategically located research sites in Kampala at Nakasero, Mukono, and Kayunga, equipped with state-of-the-art facilities to ensure high-quality data collection and participant care.
✍🏽 Laboratory Support: We are supported by a CAP-certified laboratory, ensuring adherence to the highest standards of quality and accuracy in our diagnostic and research testing.
✍🏽 Pharmacy Services: Our organization boasts a nationally certified pharmacy dedicated to managing trial drugs, ensuring safe, secure, and compliant handling of investigational medicines.
✍🏽 Quality Assurance and Community Engagement – Our Quality Assurance team ensures all research complies with regulatory standards, safeguarding participant safety, and data integrity. Our community engagement arm collaborates with stakeholders to promote transparency, trust, and community participation, enhancing the relevance and success of our research initiatives.
✍🏽 Capable Workforce Our team is equipped with relevant skills to conduct competitive clinical research and product development.
✍🏽 Resilience During COVID-19: Despite the challenges posed by the pandemic, we maintained uninterrupted clinical trial activities, demonstrating resilience and adaptability.
✍🏽 National Leadership: We are represented on the National Task Force for epidemic preparedness, contributing to national strategies for health emergencies.
✍🏽 Institutional Capacity Building: Through a research administration training grant, we have provided mentorship and support to organizations such as the College of Veterinary Medicine and Biosafety and Biosecurity (CoVAB) and the University of Mogadishu, helping establish functional Institutional Review Boards (IRBs) over the past two years.